Here’s the first claim:
Independent scientists have been unable to reproduce the positive results reported in Burzynski's studies: NCI observed that researchers other than Burzynski and his associates have not been successful in duplicating his results,[20]
It would be a monstrous quibble on the order of “It depends on what the meaning of ‘is’ is,” to say this is not a claim that there are no “researchers other than Burzynski and his associates” who have successfully duplicated his results. I suppose that technically it only claims that there exist (some) researchers who have not been successful etc, but, again, that is not the clear sense and intention of the paragraph and article. Similarly we might take the first sentence only to be claiming that there exist independent scientists who have failed to reproduce the positive results etc.
But let’s first think about the significance of this claim in terms of the efficacy of the Burzynski treatment. To me it would be interesting to know just what Dr. B’s results have been. And his associates. What were his trials? How many people did he treat? How sick were they to start with and to end with? Where did the patients come from? What exactly did he use to treat them? How statistically significant were his results? On a similar train of thinking, I’d wonder the same about those independent medical people who failed to reproduce his results. Who were they? How many independent trials did they conduct? Who were their patients? How sick? How many of them? How statistically significant? Were these scientists independent of Burzynski perhaps dependent on someone else? Would it matter if they were employed by pharmaceutical corporations with a financial interest in cancer treatments? Would it matter if they were employed by a university or the NIH but had some present or potential financial interest in the outcome of their trials?
The sentence in Wikipedia we are looking at does not even attempt to convey the beginning of a summary of that kind of information, so let’s look at the footnoted source. This is the web link: http://www.cancer.gov/about-
Well, that was easier reading than I thought it might be. Long, though.
My main conclusions:
- Unbelievably, the Wikipedia authors (anonymous) ARE doing the monstrous quibble thing. (I return to this below.)
- The Wikipedia source (cancer.gov – NCI in HIH) uses 15 clinical trials (while sourcing 19) with a wide range of results, mostly modestly positive, one entirely negative. Here is a typical positive report summary (more not given in the source):
A phase II study also conducted by the developer and his associates at his clinic reported on 12 patients with recurrent and diffuse intrinsic brain stem glioma. Of the ten patients who were evaluable, two achieved complete tumor response, three had partial tumor response, three had stable disease, and two had progressive disease. Patients ranged in age from 4 to 29 years. Treatment with escalating intravenous bolus injections of antineoplastons A10 and AS2-
The negative report is from the Mayo Clinic.
A multicenter phase II study conducted by the departments of Oncology and Neurology at the Mayo Clinic attempted to assess the pharmacokinetics, toxicity, and efficacy of antineoplastons A10 and AS2-
- Of the 15 studies used in the Wikipedia source under the heading “Human/Clinical Studies,” all but 5 are from Burzyinski. One is the Mayo Clinic study quoted in the source summary just above this, and the other 4 are from Japan with 3 different lead authors. From video in the Eric Merola documentary, I know that one author states directly that he and his associates are independent of the Dr. B Clinic.
The source summarizes its presentation of the 15 studies with this single paragraph. (My emphasis:)
To date, no randomized controlled trials examining the use of antineoplastons in patients with cancer have been reported in the literature. Existing published data have taken the form of case reports or series, phase I clinical trials, and phase II clinical trials, conducted mainly by the developer of the therapy and his associates. While these publications have reported successful remissions with the use of antineoplastons, other investigators have been unable to duplicate these results [10] and suggest that interpreting effects of antineoplaston treatment in patients with recurrent gliomas may be confounded by pre-
Notice that while the source Wikipedia uses (NCI in NIH) knows and states that case reports and series as well as phase I and phase II clinical trials conducted mostly by the developer but also by some Japanese medical scientists “have reported successful remissions with the use of antineoplastons,” Wikipedia does not let us know this. Indeed it denies the Japanese scientists when it says that “NCI observed that researchers other than Burzynski and his associates have not been successful in duplicating his results.” In this way Wikipedia hides the fact of the positive Japanese reports.
But wait, there’s more. Also hidden is the fact that NCI only knows of one study that fails to duplicate his results. I know this from reading the entire source and noticing that that is in fact the only such negative they mention. I’ve quoted their summary of the study just above. It’s not much of one, using only 9 subjects. Here’s the abstract on PubMed. The Mayo people state this in their abstract: “Although we could not confirm any tumor regression in patients in this study, the small sample size precludes definitive conclusions about treatment efficacy.”
Perhaps the source (NCI) actually knows of other studies that have failed to duplicate the Texas and Japanese results but is not telling us about them. Not likely, given how hard they worked to find what they did find. In addition, in the history section of their long report, they even explain why there are no others, at least in this country:
From 1991 to 1995, the National Cancer Institute (NCI) initiated phase II clinical trials of antineoplastons A10 and AS2-
Now those conclusions above derive only from my looking at the first claim Wikipedia’s anonymous authors make and the source they give to substantiate their claims.
(I want to emphasize that I am using the very source Wikipedia cites to defend their claims to show those claims to be false. Wikipedia says, “Independent scientists have been unable to reproduce the positive results reported in Burzynski's studies.” But the source lists four independent Japanese studies that do reproduce the positive results reported in the Burzynski studies.)
that do reproduce the positive results reported in the Burzynski studies.)
It is naturally appropriate to wonder if there might be evidence available from another source. Any sincerely inquiring person would look around. I naturally thought to see if Eric Merola could offer anything on the topic of others being able to duplicate Dr. B’s results. The answer is yes in 9 smaller and one very big way.
Four of the 9 small ways are acknowledged in the NCI=NIH Wikipedia source we have been examining on this page. They are the Japanese studies mentioned. The other 5 are given in the right margin as links to their Pub-
For the record, the Wikipedia source (as of December 7, 2015) states that it was last updated on July 7, 2015. The “big one” study was published March 19, 2015. Of course, it is quite possible that the update then was minor and that whoever did it was not even qualified to amend the page’s study and listing of “Human/Clinical Studies.” As it stands, though, the Wikipedia source is missing important current information.
The 2015 trial is important because it reports a very positive conclusion to a phase II prospective and randomized-
I place so much emphasis on this #10 because the RC (randomized control) trial is often called the gold standard of medical trials and I am not aware of any other RC’s in the antineoplaston field of research. At least none involving prospective human patient studies. Merola makes much of these results, as does the Japanese doctor (Hideaki Tsuda) in the Merola interview. See above or in right margin for link.
Furthermore, in the next paragraph of the Wikipedia article itself we find reference to the RC as highly significant:
There is no convincing evidence from randomized controlled trials in the scientific literature that antineoplastons are useful treatments of cancer, and the U.S. Food and Drug Administration (FDA) has not approved these products for the treatment of any disease.[10]
Reference [10] is to a subunit of the same NCI source we have been considering on this page. It is out of date and makes no reference to the Big One I am referencing here.
As for the details of the 2015 study, they must be scrutinized carefully and obviously require follow-
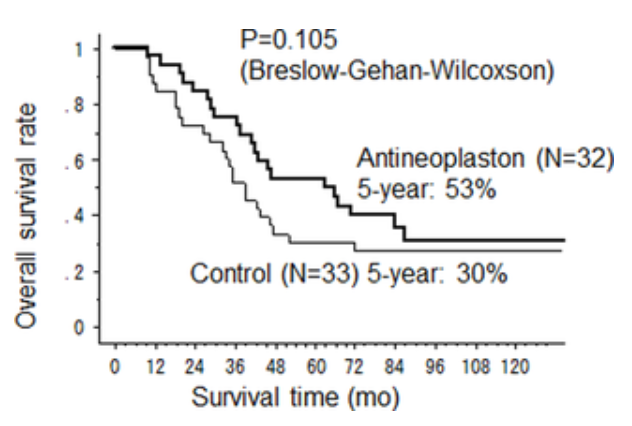
The survival time in question is (I think ) 120 months, 10 years after liver operations had been conducted for colon-
) 120 months, 10 years after liver operations had been conducted for colon-
The endpoint the scientists set themselves at the outset of the trials was something called cancer-
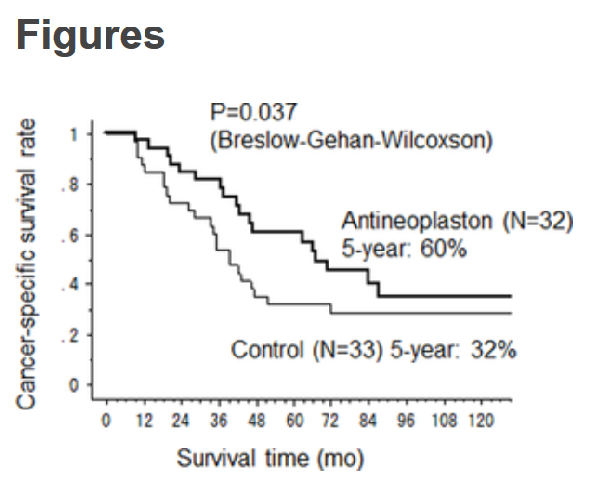
As a final observation on the accuracy and trustworthiness of this first claim in the Wikipedia Efficacy section, I must note the factual inaccuracy and sophistically misleading nature of the last clause quoted at the top of this page:
NCI observed that researchers other than Burzynski and his associates have not been successful in duplicating his results.
That observation is, of course, nowhere to be found in the source for the obvious reason that the source includes the four Japanese studies already mentioned. What the source does say is given in the summary paragraph I quote above, where I have bolded it:
While these publications have reported successful remissions with the use of antineoplastons, other investigators have been unable to duplicate these results [10]
Footnote 10 refers to the one Mayo study already mentioned. This study of nine patients is the only study the NCI was able to produce that did not confirm or tend to confirm the Burzynski results when they found four Japanese studies (other investigators) that did confirm. The clear enough intention of the NCI report, which is the Wikipedia source, is to state that certain other investigators (namely those who authored the Mayo study) were unable to duplicate the Dr. B results. But the even clearer intent of the Wikipedia authors is to state that no other researchers (than Dr. B and his associates) have been able to duplicate the results. To take their statement in any other sense is to do the Clinton dodge – “It depends on what the meaning of ‘is’ is.” And even if we acknowledge that such a meaning is possible for their sentence, we know they have hidden from the reader the four Japanese studies that independently duplicated the Dr. B results.
From my study of this first claim the anonymous Wikipedia editors make, I draw the conclusion that they are deliberately hiding good evidence in support of the Burzynski antineoplaston cancer-
This is a panel. Close it by clicking somewhere not on it. May take two clicks.
May include gaphics and hyperlinks.
For the 15 trials, see Table 2 Antineoplastons Clinical Trials in the source. They are referenced under the heading “Human/Clinical Studies.” The other four sources are used elsewhere in the summary.
As for why the Americans were unable to find participants while the Japanese were, see the first Eric Mercola documentary on Burzyniski. This also documents the reasons for the failure of the Mayo tests, which amount to their violating their agreement with Burzyinski to use his treatment protocol rather than their own.
The other 5:
The big one:
2015. See also the Merola interview with Hideaki Tsuda, one of the main investigators in the Japanese studies, including this one.
And, after all, we have the unequivocal thesis statement in the opening paragraph: “there is no evidence of the clinical efficacy of these methods.”
| Spirit Spiritual |
| Notes for Friends |
| Personal but not Private |
| Tragedy and Grief |
| My Spirit Adventures |
| Spirit Sources |
| Why we celebrate Thanksgiving |
| Where Shannon Is |
| Nurse Brown's Experience |
| Noreen Dies in the Holocaust |
| Love Consciousness |
| Baseline What a Whisper Sounds Like |
| Who Came Calling? |
| Memorial Day |
| An Earlier Day |
| Transcript of the Warner tirade |
| SHELDRAKE-Shermer 1st |
| SHERMER-Sheldrake 1st |
| SHELDRAKE-Shermer 2nd |
| SHERMER-Sheldrake 2nd |
| SHELDRAKE-Shermer 3rd |
| SHERMER-Sheldrake 3rd |
| The Jaytee Issues |
| Note 1 |
| Bob musings over this |
| Seeing with Your Tongue |
| Thinking About These Things |
| Dr. Burzyinski |
| Dr Gundry - Plant Paradox |
| Dr Macchiarini - Stem Cells, Artificial Transplants |
| The Wikipedia article |
| First Claim |
| Flowers my neighborhood |
| Where we were once in India |
| Family |
| Precession of the Equinoxes |
| Self-doubt |
| Bastard Toadflax |
| Butter and Eggs |
| Pink Evening Primrose |
| in situ Pink Evening Primrose |
| Jane Goodall Wildlife Forests Food |
| Christmas 2019 |
| Transcript of the Warner tirade |
| SHELDRAKE-Shermer 1st |
| SHERMER-Sheldrake 1st |
| SHELDRAKE-Shermer 2nd |
| SHERMER-Sheldrake 2nd |
| SHELDRAKE-Shermer 3rd |
| SHERMER-Sheldrake 3rd |
| The Jaytee Issues |
| Note 1 |
| Bob musings over this |
| Seeing with Your Tongue |
| Thinking About These Things |
| Dr. Burzyinski |
| Dr Gundry - Plant Paradox |
| Dr Macchiarini - Stem Cells, Artificial Transplants |
| The Wikipedia article |
| First Claim |
| Dr. Burzyinski |
| Dr Gundry - Plant Paradox |
| Dr Macchiarini - Stem Cells, Artificial Transplants |
| The Wikipedia article |
| First Claim |
| The Wikipedia article |
| First Claim |
| First Claim |

